AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Co2 molecular geometry11/16/2023  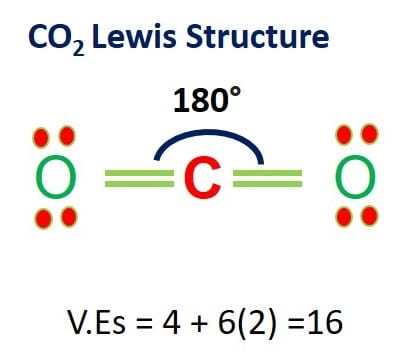 
Due to these repulsive forces between the valence shell electron pairs, the CO2 molecule acquires a linear shapeto keep the repulsion at the least.\): Water molecule. As a result, there are no lone pairs of electrons, but bonding pairs of electrons also repel each other. Here in CO2, both Oxygen atoms form sigma bonds with the central carbon atom and complete their octet. The molecular Geometry of any compound is based on the arrangement of atoms, electron pairs, and bonds. Co2 molecule vector images are visuals that show the molecular structure of carbon dioxide, one of the most abundant greenhouse gases, responsible for global. Remaining electrons in the p-orbitals in the Oxygen atom form pi bonds.Īs sp orbitals are hybridized to form the bonds, CO2 has an sp hybridization. These two hybridized orbitals overlap with the two p-orbitals of the Oxygen atom that results in the formation of sigma bonds. The molecular geometry is the shape of the molecule. The electron-pair geometry provides a guide to the bond angles of between a terminal-central-terminal atom in a compound. In contrast, the Oxygen atom hybridizes to form three sp2 hybrid orbitals. Molecular geometry is the name of the geometry used to describe the shape of a molecule. Here the 2s orbitals and one of the p-orbitals will hybridize to form 2 sp orbitals. In its excited state, the atom’s electronic configuration becomes 1s2 2s1 2p3, so now every p-orbital of the atoms has one electron each. Total number of valence electrons in the molecule = 16 Valence electrons in Oxygen: 6*2 = 12 ( as there are two Oxygen atoms in the molecule, we will multiply it by 2) The molecular geometry of any compound can be determined by the VSEPR theory. 
This liquid is used in electroplating, mining, and as a precursor for several compounds. Represented by the chemical formula, HCN is one of those molecules that has an interesting Lewis structure. To know the bond formation and the arrangement, let’s go through the valence electrons of all the atoms in the molecule. 15 May Hydrogen Cyanide is a colorless, flammable, and poisonous chemical liquid. Two Oxygen atoms are located on the terminals where both these atoms share electrons and form bonds with the central Carbon atom. In CO2, the Carbon atom is in the central position as it is the least electronegative atom in the molecule. Now that you know how the Lewis structure is drawn and its uses let us quickly look at the CO2 Lewis structure. Such structure helps in understanding the arrangement of atoms along with the electrons participating in the bond formation. Drawing lines represent the bonds formed in the molecule. These valence electrons are represented by drawing dots around the individual atoms, hence the Lewis dot structure. Lewis dot structure is a pictorial representation of the arrangement of the valence shell electrons in the molecule. To know the lewis structure of CO2, one should first understand what precisely the Lewis structure is. total valence electrons in SO42- 61 + 64 +2 32. Sulfur and oxygen both belong to the same group in the periodic table ( the chalcogen family) and have six valence electrons each. For sulfate ions, we have one molecule of sulfur and four molecules of oxygen. Please purchase a subscription to get our verified Expert's Answer. Step 1: Count the total number of valence electrons present in the molecule/ion. This structure helps in knowing the arrangement of electrons in the molecules and the shape of the molecule. What is the molecular geometry of carbon dioxide, CO2 A) tetrahedral B) trigonal pyramidal C) bent D) linear E) not enough information 10:49 AM Earl Stokes Verified Expert 6 Votes 8464 Answers This is a sample answer. One needs to know the Lewis structure in order to understand the molecular geometry of any given molecule. Name of molecule Carbon Dioxide ( CO2) No of Valence Electrons in the molecule 16 Hybridization of CO2 sp hybridization Bond Angles 180 degrees Molecular Geometry of CO2 Linear CO2 Lewis Structure 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
